Unlocking Efficiency with Track & Trace Software and Supply Chain Visibility
- Reduce Cost
- Eliminate Complexity
- Improve Visibility
- Enhance Control
- Ensure Compliance
Introducing DIAMIND Sentry: The Next Generation of Error Handling
Delivering a supply chain solution with real-time data, end-to-end traceability, environmental monitoring, and compliance. Explore what we can do for you.

Empowering our customers to protect product, profit, people, and planet.
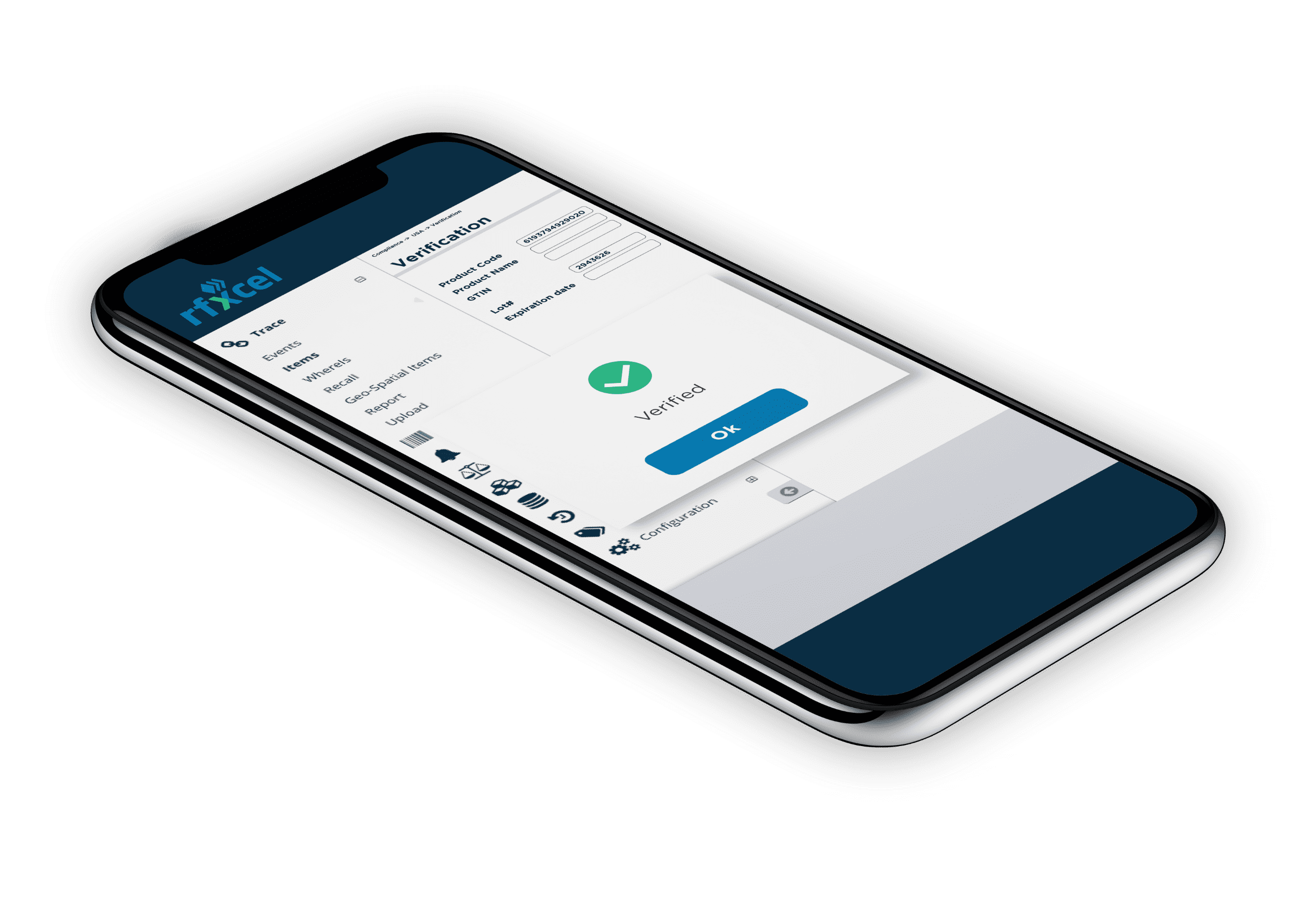
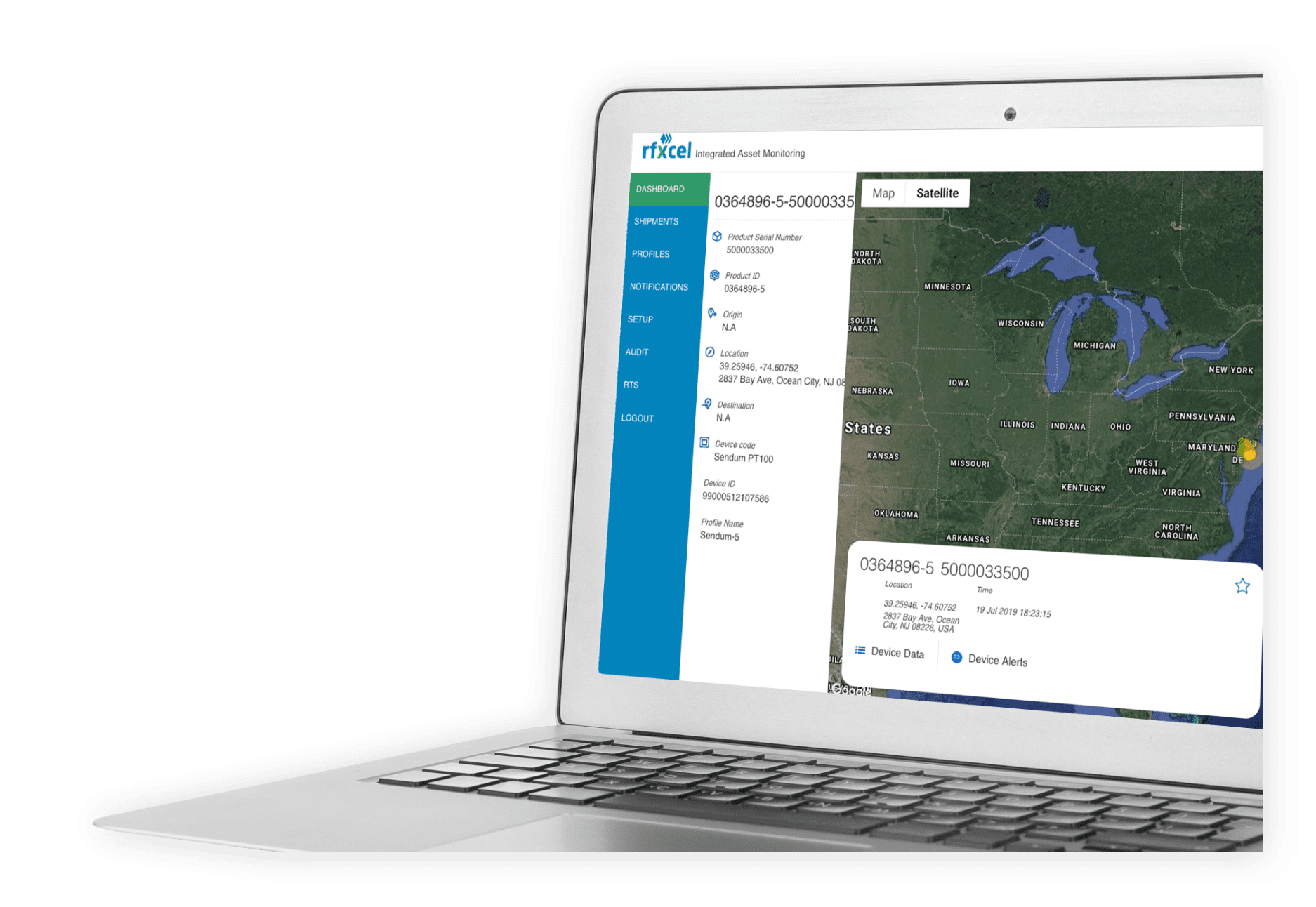
Complete Supply Chain Visibility & Security
Deliver the right product to the right place at the right time with total visibility along the way.

Protect your supply chain with rfxcel Traceability System.
rfxcel Track and Trace Solutions
See how customers are succeeding with rfxcel
Ready to see what rfxcel can do?
Latest from rfxcel

A LinkedIn Live Recap Featuring Julien Faury at Antares Vision Group EU Hub alerts now create measurable compliance risk for…

New York, NY – November, 2025 – Antares Vision Group - a global leader in traceability and quality…

A LinkedIn Live Recap Featuring Alex Colgan at Ledger Domain and Herb Wong at Antares Vision Group Watch the Full…